A Love Letter from Texas Scientists to the Periodic Table
We're celebrating the 150th anniversary of the periodic table. Join us as we tour the cosmos, from the microscopic to the telescopic, with four scientists studying the role of four elements—zinc, oxygen, palladium and gold—in life, the universe and everything.
Emily Que is a chemist who helped capture, for the first time on video, zinc fireworks that burst from an egg when it's fertilized by sperm. Astronomer Michael Endl is searching for chemical signs of life in the atmospheres of exoplanets. Kate Biberdorf (a.k.a. Kate the Chemist) found new ways to speed up chemical reactions using palladium. And physicist Aaron Zimmerman explains why the gold in your jewelry was probably forged in an ultraviolent explosion billions of years ago.
Show Notes
Watch Kate the Chemist blow things up in honor of National Periodic Table Day.
Hear more about the first neutron star collision captured by a gravitational-wave detector.
In honor of the publication of Dmitri Mendeleev's first periodic table in 1869, the United Nations has declared 2019 the International Year of the Periodic Table.
Music for today's show was produced by: Podington Bear, Chris Zabriskie and Dresden the Flamingo.
Photo of periodic table cupcakes by Vivian Abagiu.
TRANSCRIPT
Emily Que: You know, I don't know if I have one favorite element, but I will just say I'm an inorganic chemist, so metals really excite me.
Michael Endl: The most important chemical element in this respect is oxygen. So when we look at the history of life on Earth, oxygen is a key component.
Aaron Zimmerman: Do I have a favorite element? There are so many good elements. Um. [exhales, laughs] I never really thought about it. Certainly, carbon is an element we're all grateful for and for being able to exist.
Marc Airhart: Do you have a favorite element?
Kate Biberdorf: Uh, yes. I love palladium. I've worked with palladium forever. I think it's such a beautiful element.
MA: This is Point of Discovery and that was chemist Emily Que, astronomer Michael Endl, physicist Aaron Zimmerman and chemist Kate Biberdorf. They're all scientists at the University of Texas at Austin.
MA: As you probably know, this is the hundred and fiftieth anniversary of …
KB: THE PERIODIC TABLE!!!
MA: [sings: dah-dah-dah daaaaaaah]
MA: Chemistry's periodic table boils down the important information about all of the chemical elements that make up the universe into one sleek little package. Today, we'll explore what the table's 118 elements mean for our lives, for our relationship to nature and for our understanding of the universe. We'll take a closer look at what scientists are learning in relation to four elements: zinc, oxygen, palladium and gold.
MA: First up, Emily Que is a chemist who has studied element 30 on the periodic table and what it means for reproduction. That element is zinc, found in trace amounts in the human body and in all living things on Earth. Que and her team are unravelling the ways that zinc is important in fertility.
EQ: Instead of doing a super-invasive procedure to determine if an embryo is viable, you could measure the zinc that is released.
MA: Que showed me a video she and some colleagues made of what happens with zinc, at the dramatic moment when a mouse sperm fertilizes a mouse egg.
MA: It starts with this green ball. So what is that?
EQ: So the green ball is actually an egg cell and we've put a dye in that egg cell that basically labels the zinc on the inside of the cell. The egg cell is storing zinc in these little pockets, little packages at the cell surface. When the egg is fertilized, the egg releases the zinc from these packages and you see the zinc release in this red color, which represents another dye that we use to look at zinc.
MA: Oh! And all of a sudden it just changes color.
EQ: Yeah! So basically, what we saw there, right before it's fertilized, we have all of those green labeled zincs inside of the cell, and then once it's fertilized then it sort of bursts outside of the cell and creates that other color.
MA: That's really amazing.
EQ: Basically the idea is the egg maybe is releasing zinc and then preventing more sperm from fertilizing it. Because we only want one sperm to fertilize one egg. Otherwise, that isn't a viable embryo. So we think that that may be one role that zinc is playing in fertilization.
EQ: And there seems to be some relationship between how big that zinc spark is and whether that embryo will go on to become a viable living creature.
MA: Que believes one day, zinc measurements could become a standard part of helping couples struggling with fertility. Following in vitro fertilization, doctors would be able to easily assess whether eggs were successfully fertilized or likely to produce viable embryos based on how much zinc was released. Beyond such practical purposes, there's a kind of poetry in the zinc-spark discovery.
MA: Just the idea of sperm and egg meeting and then there are fireworks, it sort of sounds like two humans meeting, and you know, there are fireworks. It's like the way we talk about love, right?
EQ: Exactly. It's really pretty striking how, I don't know, anthropomorphized or whatever you want to call it, that really it's a chemical, a biochemical event, right? But yeah it really has some powerful emotional connections with what we as humans do.
MA: Now let's shift from the microscopic to the telescopic, with a look at chemical element number 8—oxygen. According to astronomer Michael Endl, oxygen could provide signs that we are not alone in the universe. He studies planets outside of our solar system—known as exoplanets. As large new telescopes become available over the next decade, Endl believes we'll start to measure chemicals in the atmospheres of some of these planets.
ME: If you could observe an earth-like planet that has for instance earth-type life on it, we would be able to see the signature of oxygen in our atmosphere. And the thing about this is, why would we think this is a biosignature? Oxygen is a highly reactive element, it combines quickly with all kinds of other stuff. Our atmosphere is in something that we call chemical disequilibrium – meaning it's not in a stable state – so you have to replenish the oxygen in our atmosphere continuously. So if a certain catastrophe happened and if all life vanished from one day to the next, the oxygen would quickly disappear – because it reacts with the rocks and other stuff. The normal, natural rust that you see is oxygen reacting with iron for instance. So this is what we're looking for – we're basically looking for an atmosphere that the chemicals that are there shouldn't be there.
MA: Endl says you'd have to find more than just oxygen to feel confident that a planet has life.
ME: What we realized over the past few years is a really good biosignature is always a combination of several things. So for instance, you have to get the temperature and density and pressure of the atmospheres correct – for this you look at other gases, other elements in the atmosphere.
MA: So that would strengthen the case if you had a combination of different chemical signatures together that would indicate life and would be harder to explain in another way.
ME: Right. For instance, methane is the next thing that comes to mind when it comes to biologically produced gases in our atmosphere.
MA: In the end, we may never be 100 percent certain a particular planet has life.
ME: Maybe in 10 or 20 years, we'll have a list of the 10 best planets where each of these planets has a probability associated with it that what we see is produced by life.
ME: If I would need to bet money, this will be the first evidence we have of extraterrestrial life. It will not come, like in the movie Contact, by a radio message. I would bet my money on a biosignature from a planet that is alive, even if it's just microbes and that would be the most spectacular discovery made in human history.
MA: Before Kate Biberdorf, a UT Austin faculty member whose chemistry demonstrations are legendary, became known for her explosive "Fun with Chemistry" shows, she spent years researching element number 46—palladium—as a catalyst. A catalyst is something that helps speed up chemical reactions. Instead of taking 10 hours for a reaction to happen, maybe you can do it in an hour. And if time is money, then catalysts are very valuable.
KB: So I was given palladium and to study all these different palladium catalysts to see which one made their reactant turn into a product. For about 6 to 7 years, I worked with palladium straight. I think it's such a beautiful element, and I love how it's such a good catalyst and it's affordable.
MA: This is where the periodic table comes in—palladium is in the same column as platinum. Platinum is a really great catalyst—but it's also very rare and expensive. Thanks to the periodic table, we know that palladium has similar properties to platinum. And it's a lot cheaper.
KB: So I know that everything I publish, all my research could be done in a lab, a school that maybe has 1,000 or 2000 kids, they can afford this catalyst. Had I been working with platinum, these small liberal arts schools could never be able to replicate my work. And so for me, it's really important.
MA: Okay, so palladium, if you could be any element ...
KB: Oh, that's different. That's a different question. I love working with palladium 100 percent., the mechanisms how the catalyst goes together, the puzzle aspect, that's so fascinating to me. But in terms of which one would I want be? That would either be hydrogen or helium -- and the reason being because those are gases and they are very, very light, so they would fly. I'm a big superhero fan, so I've always wanted to fly, so I'd have to be hydrogen or helium. And if I had to pick, it would have to be hydrogen because hydrogen is explosive, so if you set it on fire, you're going to have a huge fireball, whereas helium just goes POP and it's lame. So I think hydrogen.
MA: Wow. Flying and explosive?
KB: That's definitely me.
Listen to the Episode
MA: Finally, we turn to the heavy side of the periodic table. That's where we find element 79—gold. Physicist Aaron Zimmerman is part of LIGO – a "big science" project that has been listening for rumbles in the universe that make time and space quiver like a guitar string: what are known as gravitational waves. In August 2017, LIGO's gravitational-wave detectors caught for the first time two super-dense objects called neutron stars smashing into each other.
AZ: What we think happened was we had these two super dense objects, let's say 11 kilometers across and with more mass than our sun in them, so densities that you can't imagine. And they're whipping around each other at ten percent the speed of light, the kinds of speed you can't even imagine as they whirl and whirl around each other and then finally they collide together and there's a blast of material that gets squirted out of where the two crash together.
MA: The explosion released a blue-white fireball of unimaginable heat and matter. It was so hot and dense that it created some of the heaviest elements in the universe like platinum, uranium and gold.
AZ: And it turns out by observing this radioactive fireball—how brightly it glowed, how hot it was, how quickly it faded—astronomers were able to estimate how much material was in that fireball coming off this collision. And from that they were able to deduce that there were a lot of heavy elements produced. In fact, many times earth's mass of gold was produced. I was just looking at a paper trying to get a better number for this call and between 15 and 70 earth masses is what one team's estimate for the amount of gold that was produced in this event.
MA: Good grief! That's a lot of gold.
AZ: And that's just one component. In addition, this was also making platinum, uranium, all of these elements at the top of the periodic table, basically were being manufactured in this fireball.
MA: Astronomers think that neutron star collisions like this—as well as some exploding stars called supernovas—created most of the heavy elements in the universe.
AZ: It would have been mixed in with all the gasses and dust of the galaxy and eventually when our own solar system was born out of the collapse of some cloud of gas, the trace bits of gold and so on that accreted onto the earth and that we can find today would have come from something exactly like one of these collisions.
MA: So, if you have a gold ring on, you may have on your finger something that was created in a massive explosion billions of years ago?
AZ: Yes. Evidence is pointing to the fact that the gold on your finger came from some extremely violent explosion or collision back in the earlier parts of our galaxy's history.
MA: Point of Discovery is a production of the University of Texas at Austin's College of Natural Sciences. Our senior producer is Christine Sinatra. Visit us on the web at pointofdiscovery.org. While you're there, you can watch a video of zinc-spark fireworks, listen to an earlier podcast featuring Kate the Chemist and see a periodic table showing how each element was created. You'll also find notes about the music you heard in this episode. Again, that's pointofdiscovery.org. I'm your host and producer Marc Airhart. Thanks for listening!
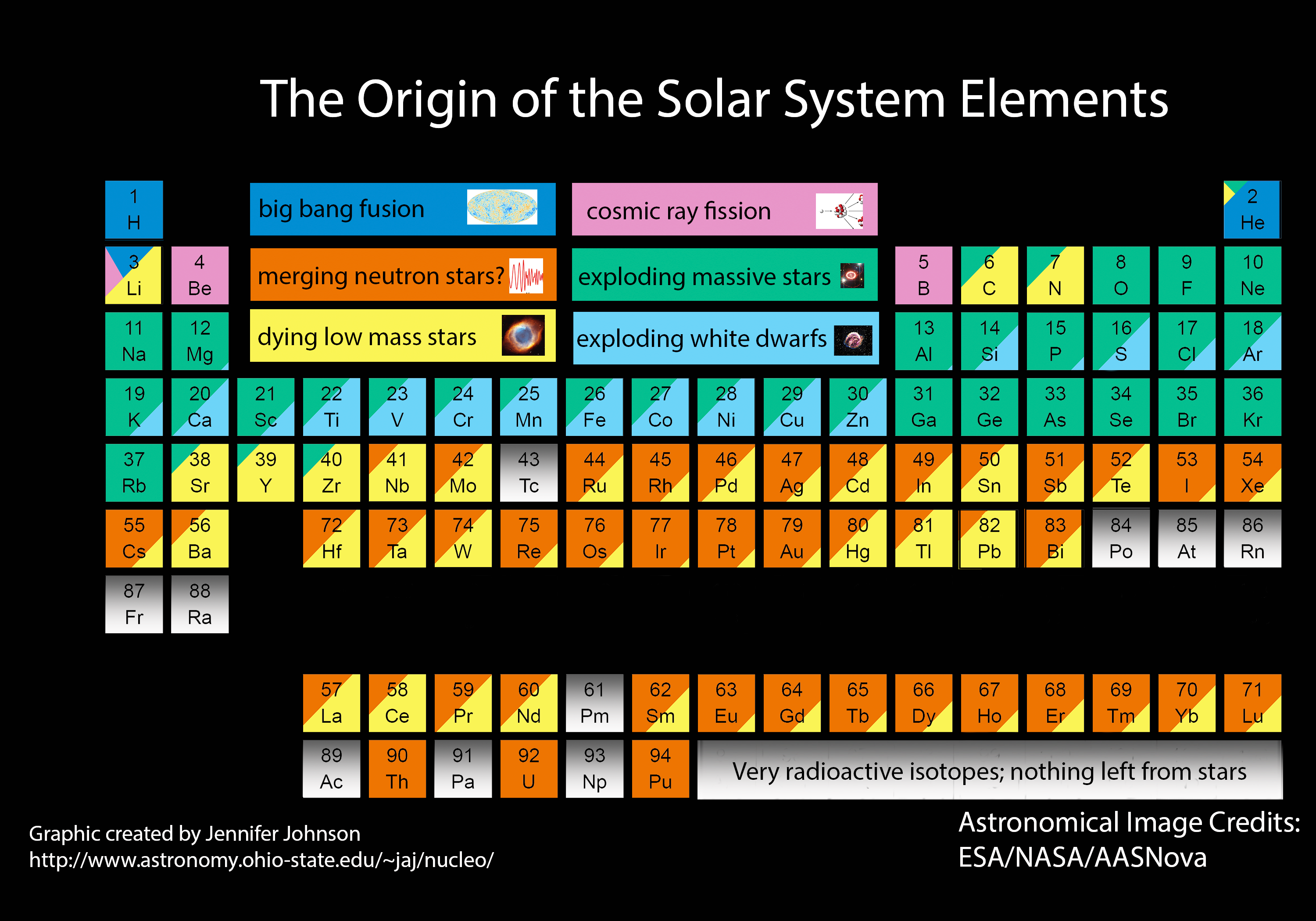
Origin of the Elements in the Solar System by Jennifer Johnson is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License. Initial information on the Periodic Table color-coded by stellar origin can be found in this blog post: http://blog.sdss.org/2017/01/09/origin-of-the-elements-in-the-solar-system/



