Researchers Identify Potential New Target for Treating T Cell Leukemia
A team led by Lauren Ehrlich found a link between myeloid cells and the cancer that often strikes children.
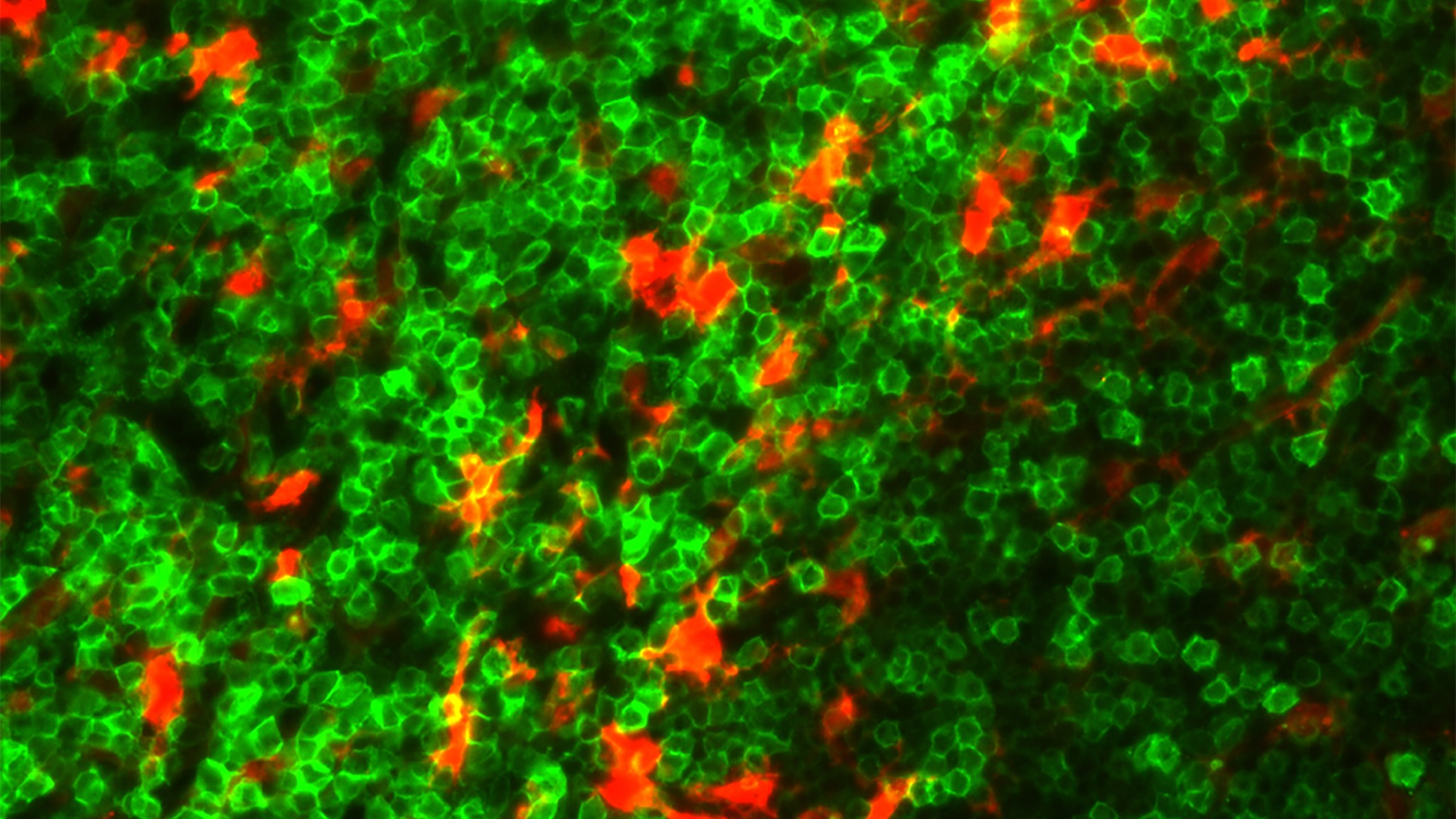
T cell acute lymphoblastic leukemia cells (green) and myeloid cells (red) in the spleen of leukemic mice. Credit: Ryan Humphrey/University of Texas at Austin.
More than a thousand people in the U.S. each year are diagnosed with a kind of blood cancer called T cell acute lymphoblastic leukemia (T-ALL), and about half of them are children. Existing chemotherapies against T-ALL can add years to a patient’s life but have awful side effects — including neurological toxicity, cognitive impairment and metabolic disorders. And if the cancer develops resistance, the patient’s survival rate plummets.
A research team led by Lauren Ehrlich, professor of molecular biosciences at The University of Texas at Austin, discovered that T-ALL cells have to adhere to another group of cells in the local environment, called myeloid cells, to receive a survival signal. If the signal is disrupted, they found, the leukemia cells die. If the cancerous cell connects with a myeloid cell, however, it sets off a cascade of downstream signals that can cause growth in the leukemia cell with the activation of two key proteins. When the researchers inhibited those proteins, the T-ALL cells also died, which suggests a possible new target for T-ALL therapies. Their paper about the work appears in Nature Communications.
“These findings suggest that inhibitors of cellular interactions between leukemia cells and cells in their environment could serve as promising novel therapies for T-ALL,” said Ehrlich, the senior author, who holds the L. Leon Campbell, Ph.D. Distinguished Professorship in Microbiology.
Notably, drugs targeting these pathways are currently in clinical trials to treat other kinds of cancers and have passed Phase I safety trials.
T-ALL affects developing immune cells called T cells. Ehrlich and her team discovered that T-ALL cells require direct contact with myeloid cells in order to survive. Think of the T-ALL cell as a set of bowling pins. The bowling ball (a myeloid cell) has to make contact with the first pin before anything interesting can happen. Then, as that first pin falls, it sets off a cascade of tumbling pins.
Specifically, her team found that when the myeloid cell sticks to the T-ALL cell, via cell surface receptors called integrins, it turns on a signaling pathway that involves two proteins called FAK and PYK2. When this pathway is activated, a T-ALL cell is then able to use a special growth factor produced by the myeloid cell it’s attached to, called IGF1. If the signaling pathway is not activated, the T-ALL cell is unable to use the growth factor and it dies.
“Most research on T-ALL has been focused on mutations in the genome of the leukemia cell,” Ehrlich said. “And that’s an angle that sets us apart from most leukemia research. We’re showing how the tumor microenvironment supports leukemia growth and survival.”
The researchers also found evidence that pediatric T-ALL patients with increased activation of integrin adhesion and associated FAK signaling had worse survival rates.
In patients with T-ALL, the immature T cells become cancer cells and proliferate extensively, which weakens the immune system. Because T cells travel through the blood stream, the cancer often spreads quickly throughout the body — to the liver, spleen, lymph nodes and central nervous system — making it especially difficult to treat. Notably, the Ehrlich lab found that healthy myeloid cells in multiple organs support the growth and survival of leukemia cells.
Much more work remains to determine if drugs targeting the proteins FAK and PYK2 will be safe and effective in actual T-ALL patients, including additional studies in animal models.
The study’s co-first authors are Aram Lyu, a former Ph.D. student, and Ryan Humphrey, a current graduate student, both in Ehrlich’s lab at UT Austin. Cancer cells from T-ALL patients were provided by co-author Terzah Horton, associate professor at Baylor College of Medicine/Dan L. Duncan Cancer Center and Texas Children’s Cancer Center.
This work was supported by the Cancer Prevention and Research Institute of Texas (CPRIT), the American Cancer Society and the William and Ella Owens Medical Research Foundation. Ehrlich was originally recruited to UT as a CPRIT Scholar.