Researchers Discover Key Mechanism Cells Use to Harness Energy
New study finds how cells use important nutrient.

Mitochondria in a cell. Image courtesy of Paul A. Oliphint.
Researchers at The University of Texas at Austin and The University of Pennsylvania identified the molecular transporter that enables the mitochondria of human cells to take up the vital NAD+ nutrient.
"The way that we break down nutrients into energetic units that our cells can use is at the root of cellular life," said Lulu Cambronne, assistant professor of molecular biology at The University of Texas at Austin.
Despite years of study, scientists did not know exactly how humans obtained their mitochondrial pool of NAD+. Previously, scientists had identified the biochemical transporter that moved NAD+ into plant and yeast mitochondria, but scientists weren't even sure a counterpart existed in mammalian cells.
Cambronne and her team developed unique biosensors that provided the glimpse into how human mitochondria take in NAD+. These biosensors then aided in the identification of the candidate protein, and her team provided direct biochemical evidence to designate that this newly identified protein functions as a transporter. By zeroing in on this regulatory step and identifying the key mitochondrial protein that orchestrates this process, scientists provide new hope and clues to aging and human diseases related to metabolism and mitochondrial function, such as cancer, diabetes and cardiovascular disease.
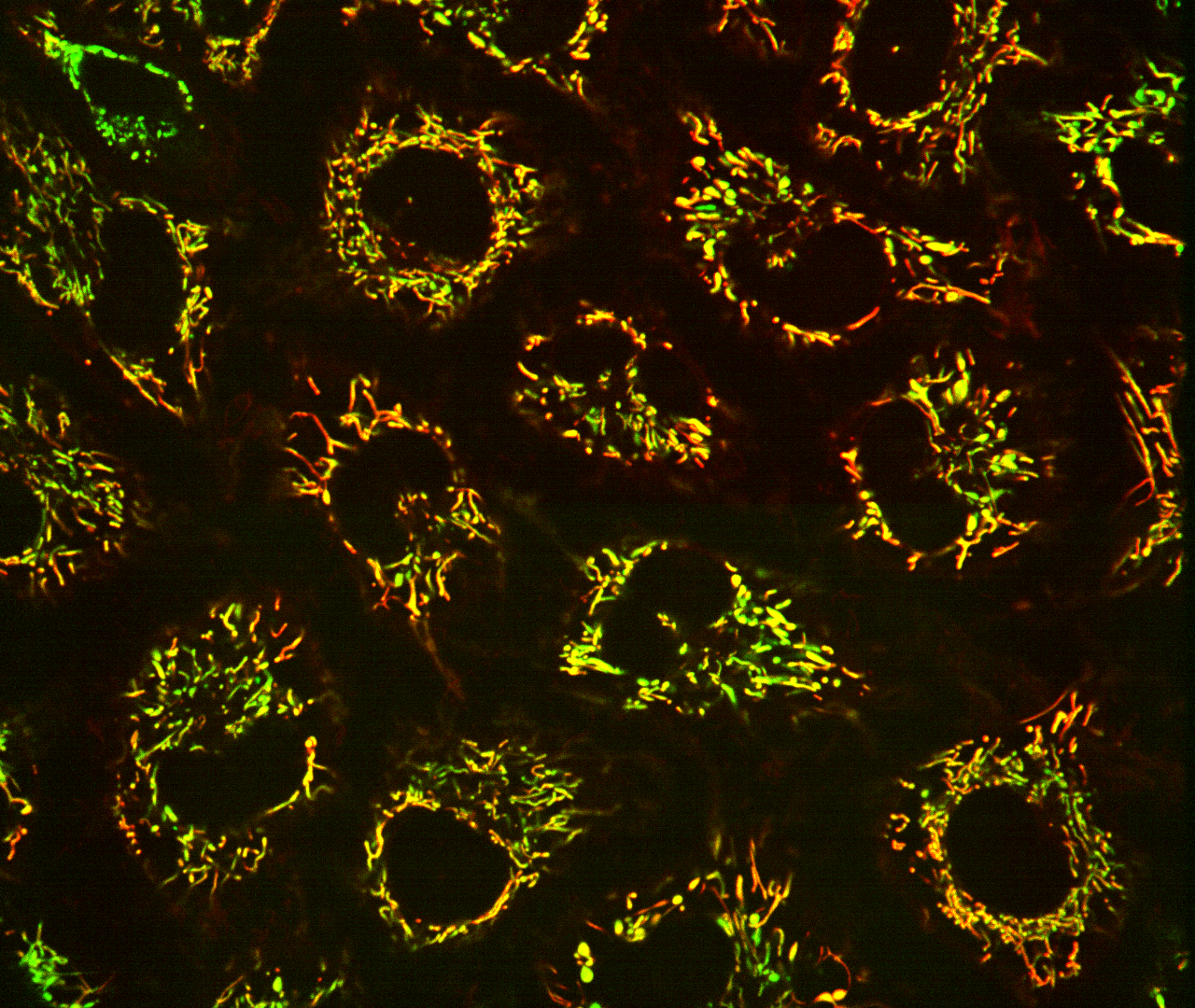
Fluorescent biosensors localized to the mitochondria of human cells reveal how the organelle controls its levels of oxidized nicotinamide adenine dinucleotide (NAD+), a molecule that when deficient is suggested to play a role neurodegenerative disorders, cardiovascular diseases and Type II diabetes. Photo courtesy Lulu Cambronne.
"This discovery means that the process of how we use energy in our cells can be regulated," said Cambronne. "When something is regulated it represents a point that can be mis-regulated in disease. It could be a new target to treat disease. We know that many diseases are brought on by metabolic processes in the cell. Being able to have a new point that we can target is exciting."
NAD+ is a derivative of niacin, also known as vitamin B3, a nutrient found in foods such as chicken, turkey, mushrooms and peanuts.
"This is a really fundamental question that didn't have an answer for a long time," Cambronne said. "Now it does."
Jared M. Eller, Mu-Jie Lu, and Paul Oliphint of The University of Texas at Austin, Timothy S. Luongo, Caroline Perry , Marc R. Bornstein, F. Brad Johnson and Joseph A. Baur of the University of Pennsylvania, Marc Niere and Mathias Ziegler of the University of Bergen, Fabio Raith and Kai Johnsson of the Max Planck Institute, Lin Wang, Melanie R. McReynolds and Joshua D. Rabinowitz of Princeton University, and Marie E. Migaud of the University of South Alabama contributed to the research.
The research was funded by the National Institutes of Health.