New Technique Reveals Protein Production in Early Embryos
The earliest stages of developing life have been, in many ways, a black box for researchers before now.
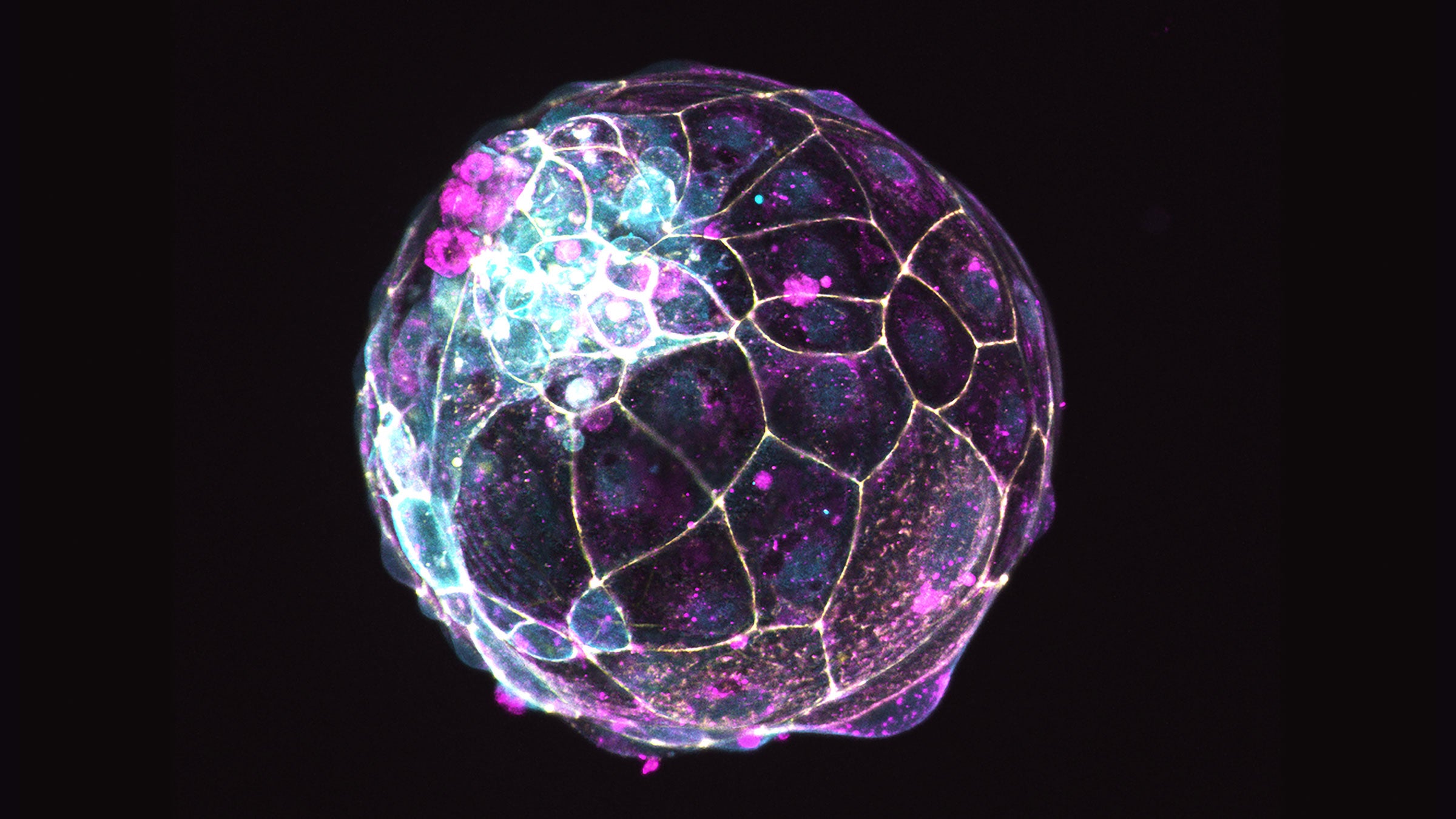
A human blastoid is an embryo model formed from stem cells that laboratories have used to study the processes of early embryo development. A new technology from UT Austin scientists, called Ribo-ITP, allows for a new way of studying early embryos and single cells as never before. Image licensed via Creative Commons (CC BY-SA 4.0). Credit: Nicolas Rivron, WikiMedia.
Scientists studying how embryos develop have had to cope with limited knowledge about what happens immediately after an egg is fertilized. An existing tool for taking a snapshot of which proteins are being made and in what abundances, called ribosome profiling, only works for samples consisting of hundreds of thousands or millions of cells.
Now, a new technology developed by a University of Texas at Austin-led team, allows researchers to study translation — the process of turning RNA into proteins — in exquisite detail in single cells and embryos for the first time. The researchers published their results in the journal Nature.
“Problems during the very early stages of development, including fertilization and shortly after, are a major cause of infertility and pregnancy loss,” said Can Cenik, an assistant professor of molecular biosciences at UT Austin and senior author of the study. “Understanding these stages in greater detail can eventually help us develop better treatments and interventions for these conditions. For example, it might help improve in vitro fertilization techniques.”
Given that translation is a fundamental process, other potential applications of the new technology exist. The advance could support the study of immunology and diseases such as cancer.
The new method, called Ribo-ITP, uses a specially designed micro-fluidic chip to isolate fragments of RNA that are in the process of being “read” like blueprints by ribosomes to produce proteins. It enables high quality and high coverage measurements of translation from single cells, a challenge that has bedeviled the field for more than a decade.
As an egg matures and immediately after fertilization, the set of proteins present is dynamically remodeled. This latest study revealed which genes are translationally regulated in these early stages.
“Fertilization is the start of a new organism’s life cycle, marking the moment when the egg and sperm combine to form a single cell — the zygote,” Cenik said. “All the complexities of an organism, whether it’s a mouse or a human, spring from this single cell. Understanding how proteins, which are the primary building blocks and functional units of cells, are regulated and function at this critical stage is key to understanding the fundamental processes of life and development.”
In addition to describing their new method, Cenik’s team reports the discovery that a protein called APC/C, plus several components of a structure involved in cell division (called the centrosome), are translationally upregulated — meaning they are produced at increased rates — upon fertilization. They also found that the abundance of proteins after an egg is fertilized is predominantly determined by the amount of their translation occurring in the egg before it is fertilized.
“Our research clarifies the crucial role of maternal translation in mammalian development,” Cenik said. “These are problems that have been studied for many decades, and our research provides the most comprehensive picture of the global contribution of different gene-expression programs to control this fundamental stage of life.”
The study’s co-first authors are Hakan Ozadam and Tori Tonn at UT Austin and Crystal Han at San Jose State University.
This work was supported in part by the National Institutes of Health, the Welch Foundation, the National Institute of Standards and Technology and the Cancer Prevention and Research Institute of Texas (CPRIT). Cenik is a CPRIT Scholar in Cancer Research.
The University of Texas at Austin is committed to transparency and disclosure of all potential conflicts of interest. University investigators involved in this research have submitted required financial disclosure forms with the university and have filed patent applications on the technology described here (63/286,531).